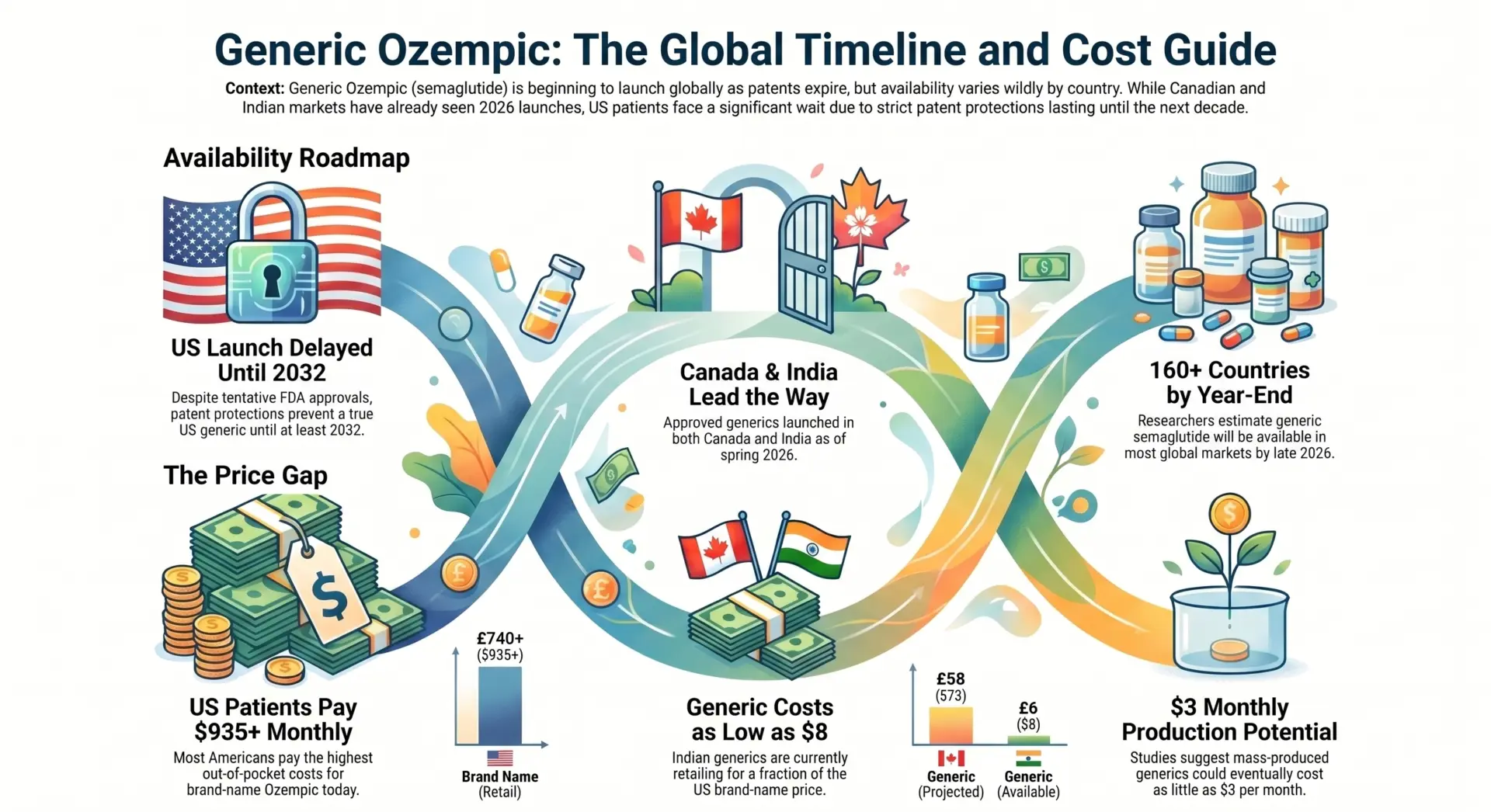
Millions of Americans pay $935 or more per month for Ozempic out of pocket, and they want to know one thing: when will a cheaper version exist? The short answer is that U.S. patients are unlikely to see an FDA-approved generic semaglutide injection before December 2031 at the earliest. Canadian patients, on the other hand, already have access to approved generics as of spring 2026. Ozempic is an injectable GLP-1 receptor agonist containing the active ingredient semaglutide, prescribed for type 2 diabetes and, increasingly, for weight loss under the brand name Wegovy.
TL;DR
- A true generic version of Ozempic (semaglutide) won’t reach U.S. pharmacies until at least 2032 because of Novo Nordisk’s patent protections.
- Canada already approved its first two generics in April and May 2026, and India has options selling for as low as $8/month.
- While you wait, brand-name Ozempic from licensed Canadian pharmacies costs a fraction of U.S. prices.
What is generic Ozempic (semaglutide)?
A generic drug contains the same active ingredient, the same dosage form, and the same strength as the brand-name original, but a different manufacturer produces it at a lower price. Generic drugs must meet the same quality and safety standards set by regulatory agencies like the FDA. Because semaglutide is a small-molecule peptide, it qualifies for traditional generic (ANDA) approval. It is not a biologic, so it does not need the more complex biosimilar pathway.
Three branded semaglutide products currently exist:
- Ozempic (injection, 0.25 mg to 2 mg weekly) for type 2 diabetes and cardiovascular risk reduction
- Wegovy (injection, up to 2.4 mg weekly) for long-term weight management in obese or overweight adults
- Rybelsus (oral tablet, 3 mg to 14 mg daily) for type 2 diabetes
Compounded semaglutide, which some U.S. patients have been using, is not the same thing as a generic. Compounded versions are mixed by compounding pharmacies and do not go through the FDA’s full review for safety, effectiveness, and quality. Dosing errors are also more common with compounded injections, since patients often need to measure doses manually from a vial rather than using a prefilled pen.
When will generic Ozempic be available in the USA?
The U.S. timeline hinges on patent expiry and FDA approval. Here is where things stand:
Patent expiry: Novo Nordisk’s core U.S. compound patent on semaglutide expires in December 2031. Additional formulation patents may extend protection into 2032.
First tentative approval: In April 2026, Apotex (partnered with Orbicular) received the first-ever FDA tentative approval for generic semaglutide injection via the ANDA pathway. “Tentative approval” means the FDA confirmed the generic meets all quality, safety, and efficacy standards, but Apotex cannot sell it yet because Novo Nordisk’s patents remain active.
Industry interest: At least 13 companies have contacted the FDA expressing interest in selling generic semaglutide.
Realistic launch: 2032 at the earliest, unless patent settlements or court rulings speed things up.
Compounding crackdown: Compounded semaglutide was widely available during the FDA-declared shortage (resolved February 2025). In April 2026, the FDA proposed excluding semaglutide from the 503B bulks list, which would shut down most remaining compounding supply.
The bottom line for U.S. patients: a true, FDA-approved generic version of Ozempic is still years away. Any product marketed as “generic Ozempic” in the U.S. today is not FDA-approved.
Generic Ozempic in Canada, already here
Canada became the first G7 country to approve generic semaglutide, and the backstory matters. Novo Nordisk missed a mandatory patent maintenance fee, causing its foundational Canadian patent to lapse in 2020. The 8-year regulatory data exclusivity window expired on January 4, 2026, and a secondary injectable-formulation patent expired in March 2026. That cleared the runway.
- April 28, 2026: Health Canada approved the first generic semaglutide, manufactured by Dr. Reddy’s Laboratories.
- May 1, 2026: Health Canada approved a second generic from Apotex, a Canadian company.
- Pipeline: Seven more submissions are currently under review by Health Canada.
- Pricing: Under the pan-Canadian Pharmaceutical Alliance, the first generic costs 75-85% of the brand price. Once a second generic enters, both drop to 50% of the brand price.
For context, brand-name Ozempic already costs roughly $147 per month in Canada, compared to $935+ in the United States. With generics now on the market, Canadian prices will fall even further.
When is generic Ozempic coming to other parts of the world?
India: Novo Nordisk’s patent expired March 20, 2026. Sun Pharma, Zydus, and Alkem have already received regulatory approval. Sun Pharma launched its generic at roughly $8/month, compared to Novo’s $100+/month in the Indian market. India is the most advanced generic market for semaglutide globally.
China: Seventeen generic candidates are in Phase 3 trials or the pre-market application stage. Launches are expected in 2026-2027.
Brazil, Turkey, and other middle-income countries: Patents are expiring throughout 2026. Generic entries are expected before year-end.
Europe (EU/UK): Extended patent terms and regulatory data exclusivity protect semaglutide until the early 2030s. EU patients will likely wait the longest among major markets.
Global projection: Researchers estimate generic semaglutide could be available in 160+ countries by the end of 2026.
A University of Liverpool study found that mass-produced generic semaglutide could cost as little as $3/month, or $28-$134 per person per year. The gap between manufacturing cost and U.S. retail price remains staggering.
What will generic Ozempic cost?
Here is what patients pay today, and what they can expect once generics reach their market:
- Brand Ozempic (USA, no insurance): $199-$499/month, depending on pharmacy and dose. The list price was $936/month in 2023 and dropped to approximately $675/month in 2026.
- Brand Ozempic (USA, with insurance): $25-$150/month, depending on plan.
- Brand Wegovy (USA, list price): Approximately $1,349/month as of 2022.
- Brand Ozempic (Canada): Roughly $147/month.
- Generic semaglutide (Canada, projected): About 50% of brand price once two generics are on the market.
- Generic semaglutide (India): Roughly $8/month
- Projected generic cost at scale: Under $3/month (University of Liverpool study).
The math is hard to ignore: a drug that costs over $1,000/month in the U.S. could be manufactured for under $40/year. U.S. patients, however, won’t see those savings until 2032 at the earliest, unless they access medications through Canadian pharmacy services.
How to prepare for generic Ozempic
You can’t buy a generic in the U.S. yet, but you can get ready:
- Talk to your doctor
Ask whether your current Ozempic dose and formulation will have a direct generic equivalent. Not every strength may launch on day one.
- Understand your insurance
Check whether your plan will prefer generics once available. Most will, and copays typically drop.
- Don’t fall for scams
The FDA has not approved any generic semaglutide for sale in the U.S. as of May 2026. Anything marketed as “generic Ozempic” in the U.S. right now is not FDA-approved. The FDA warns that unapproved semaglutide products do not go through the agency’s review for safety, effectiveness, and quality.
- Consider Canadian pharmacy options
Canadians already have approved generics, and services like PandaMeds.com offer brand-name semaglutide shipped from licensed Canadian pharmacies at savings of up to 80% compared to U.S. cash prices. Canadian pharmacies can legally provide prescription medications, including GLP-1 drugs, to U.S. customers with a valid prescription from a licensed healthcare provider.
- Monitor the FDA
Follow FDA.gov/drugs for official approval announcements.
Important: Do not stop or modify your semaglutide regimen in anticipation of generics. Semaglutide carries a boxed warning about thyroid C-cell tumor risk based on animal studies, and any changes to your treatment should be discussed with your prescriber.
PANDA10 Canada: reliable semaglutide until generics arrive
U.S. patients face a 6+ year wait for generic Ozempic. In the meantime, Canadian pharmacies already stock brand-name semaglutide at prices far below American retail.
PandaMeds.com is a reliable Canadian prescription referral service that ships prescription semaglutide (Ozempic, Wegovy, Rybelsus, etc.) directly to U.S. customers. Savings reach up to 80% compared to U.S. cash prices. All orders require a valid prescription from a licensed healthcare provider. Improper storage during shipping of injectable drugs can cause quality issues, and PandaMeds follows cold-chain shipping protocols to keep medications refrigerated in transit.
Use code PANDA10 at checkout for 10% off your first order.
Sources:
Disclaimer: This information is intended for general knowledge and informational purposes only and does not constitute medical advice. Always consult with a healthcare professional for personalized guidance.
Written by the Pandameds.com Editorial Team
Our content is created by pharmacy-trained researchers and healthcare specialists and rigorously reviewed by a diverse panel of authentic experts from the pharmaceutical and healthcare fields. This collaborative review process ensures that every article meets the highest standards of medical accuracy, reliability, and relevance.
- ✅ Authored by pharmacy-trained professionals
- 🔍 Reviewed by multiple verified experts in the pharmaceutical and healthcare niche
- 💊 Based on trusted sources including FDA, Health Canada, and peer-reviewed clinical studies
- 🔄 Regularly reviewed and updated every 90 days to maintain accuracy and trustworthiness
About Pandameds.com
Pandameds.com is a prescription referral service and help U.S. patients to access affordable and authentic weight loss medications.
Fast, Reliable Shipping to the USA!
Affordable Prescription Meds From Canada
Join our mailing list for exclusive promos, curated health content & more.
Frequently Asked Questions
Is there a generic version of Ozempic available in the U.S.?
No. As of May 2026, no FDA-approved generic semaglutide is available for sale in the United States. Apotex received tentative approval in April 2026, but Novo Nordisk's patents prevent it from launching until at least December 2031.
Is generic Ozempic available in Canada?
Yes. Health Canada approved the first generic semaglutide (Dr. Reddy's) on April 28, 2026, and a second (Apotex) on May 1, 2026. Seven more applications are under review.
Will generic Ozempic be cheaper?
Yes, significantly. In India, generic semaglutide already sells for about $8/month. In Canada, prices are expected to drop to 50% of the brand price once two generics are available. At manufacturing scale, researchers project costs as low as $3/month.
Is compounded semaglutide the same as generic Ozempic?
No. Compounded semaglutide is prepared by compounding pharmacies and does not go through the FDA's full review process. It may contain different salt forms (like semaglutide sodium or semaglutide acetate) and carries a higher risk of dosing errors. A true generic must meet the same safety and efficacy standards as the brand-name drug.
Can I buy generic Ozempic from Canada and ship it to the U.S.?
Canadian pharmacies can legally provide prescription medications to U.S. customers if the customer has a valid prescription from a licensed healthcare provider. Services like PandaMeds.com facilitate this process. Check the GLP-1 cost calculator at PandaMeds to compare your savings.
Related Blog Posts
Call Us Today!
If you have any questions, please email our support team at support@pandameds.com or call us toll-free at 1-888-862-1210.