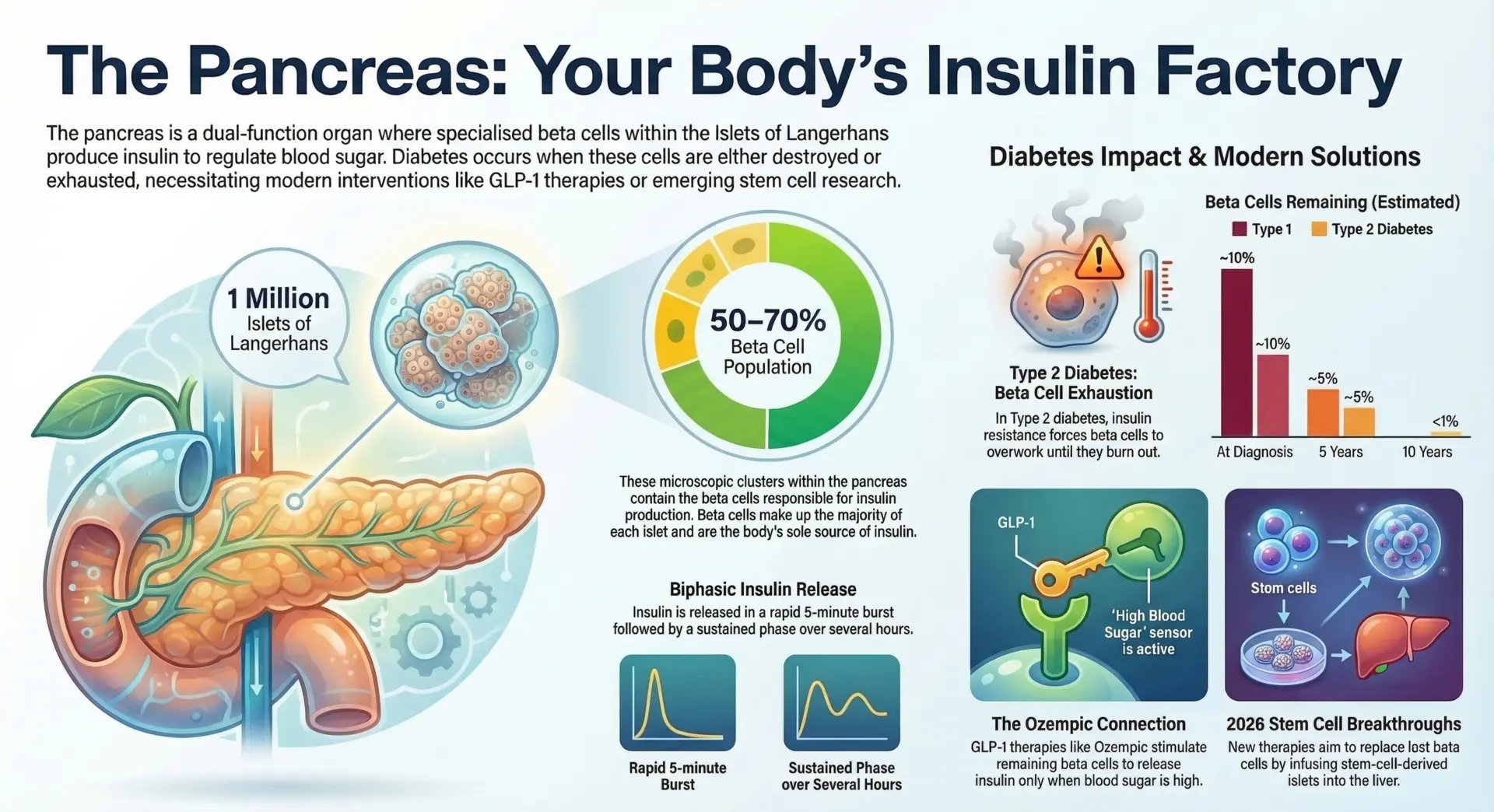
The pancreas produces insulin. This large gland, located behind the stomach, serves as both a digestive and endocrine organ. Its endocrine portion making up roughly 1-3% of the organ’s total volume contains clusters of hormone-secreting cells called the islets of Langerhans.
Within these islets, beta cells are the insulin-producing cells responsible for synthesizing, storing, and releasing the hormone insulin into the bloodstream. Insulin is an essential hormone that allows glucose the body’s primary fuel to move from the blood into the body’s cells, where it is used for energy or stored for later use.
When the pancreas does not produce enough insulin, or when the body does not use insulin properly, blood sugar levels build up. This is the fundamental mechanism behind diabetes mellitus a group of conditions that includes type 1 diabetes, type 2 diabetes and gestational diabetes.
TL;DR
- The pancreas is the organ that produces insulin. Specifically, specialized cells called beta cells, located within clusters known as the islets of Langerhans, manufacture and release this essential hormone.
- Insulin regulates blood sugar levels by helping the body’s cells absorb glucose from the bloodstream.
- When beta cells fail destroyed by an autoimmune reaction in type 1 diabetes or exhausted by insulin resistance in type 2 diabetes blood glucose levels rise, leading to diabetes mellitus.
- In 2026, GLP-1 receptor agonists like Ozempic (semaglutide) offer a powerful bridge therapy for type 2 diabetes, stimulating remaining beta cells while promoting weight loss of up to 15%.
- Meanwhile, stem cell-derived islet therapies such as Vertex’s zimislecel are advancing through Phase 3 trials with regulatory submissions expected in 2026.
Answer: Pancreas Beta Cells Produce Insulin
The pancreas is the organ that produces insulin. It is a large gland, roughly 15 cm (6 inches) long, that sits behind the stomach and is connected to the small intestine. The pancreas has two distinct functional roles:
- Exocrine function (approximately 97% of pancreas volume): Produces digestive enzymes such as lipase and amylase that break down fats, proteins, and carbohydrates in the small intestine.
- Endocrine function (approximately 1-3% of volume): Produces hormones, primarily insulin and glucagon, through cells organized in the islets of Langerhans.
The human pancreas contains approximately 1 million islets of Langerhans. Each islet is a micro-organ housing several cell types. The most important are:
| Cell Type | Hormone | Function | Diabetes Impact |
| Beta Cells | Insulin | Lowers blood glucose | Destroyed in T1D; exhausted in T2D |
| Alpha Cells | Glucagon | Raises blood glucose | Overactive in T1D |
| Delta Cells | Somatostatin | Regulates both hormones | Generally normal function |
Beta cells constitute approximately 50-70% of each islet and are the sole source of insulin synthesis in the human body. A healthy person’s beta cells produce roughly 35-50 units of insulin per day about half as a steady baseline (basal insulin) and half in response to meals (bolus insulin). This carefully calibrated output is what maintains healthy blood sugar levels throughout the day and night.
How Beta Cells Make Insulin: Step-by-Step
Insulin production is an intricate, multi-step process that begins with gene transcription inside the beta cell and ends with the release of mature insulin into the bloodstream. Here is the pathway:
- DNA → Preproinsulin: The insulin gene is transcribed into messenger RNA, which is then translated into a precursor protein called preproinsulin on ribosomes.
- Signal Cleavage → Proinsulin: Inside the rough endoplasmic reticulum, the signal peptide is removed, and the molecule folds into proinsulin with three critical disulfide bonds.
- Processing → Mature Insulin: In the Golgi apparatus, proinsulin is cleaved into mature insulin and a byproduct called C-peptide. Both are packaged into secretory granules.
- Storage → Secretory Granules: Mature insulin is stored in granules within the beta cell, ready for release when blood glucose levels rise.
Glucose-Stimulated Release: When blood glucose rises above approximately 100 mg/dL, glucose enters the beta cell and is metabolized, producing ATP. This triggers closure of potassium channels, depolarization of the cell membrane, and opening of calcium channels. The influx of calcium causes insulin granules to fuse with the cell surface and release insulin into the bloodstream.
This release follows a biphasic pattern: an initial rapid burst within 5 minutes, followed by a sustained second phase over hours. About 50% of daily insulin production is basal (overnight and between meals), while the other 50% is bolus (triggered by food intake). Insulin first travels via the portal vein to the liver, where it regulates stored glucose, before reaching the rest of the body’s cells.
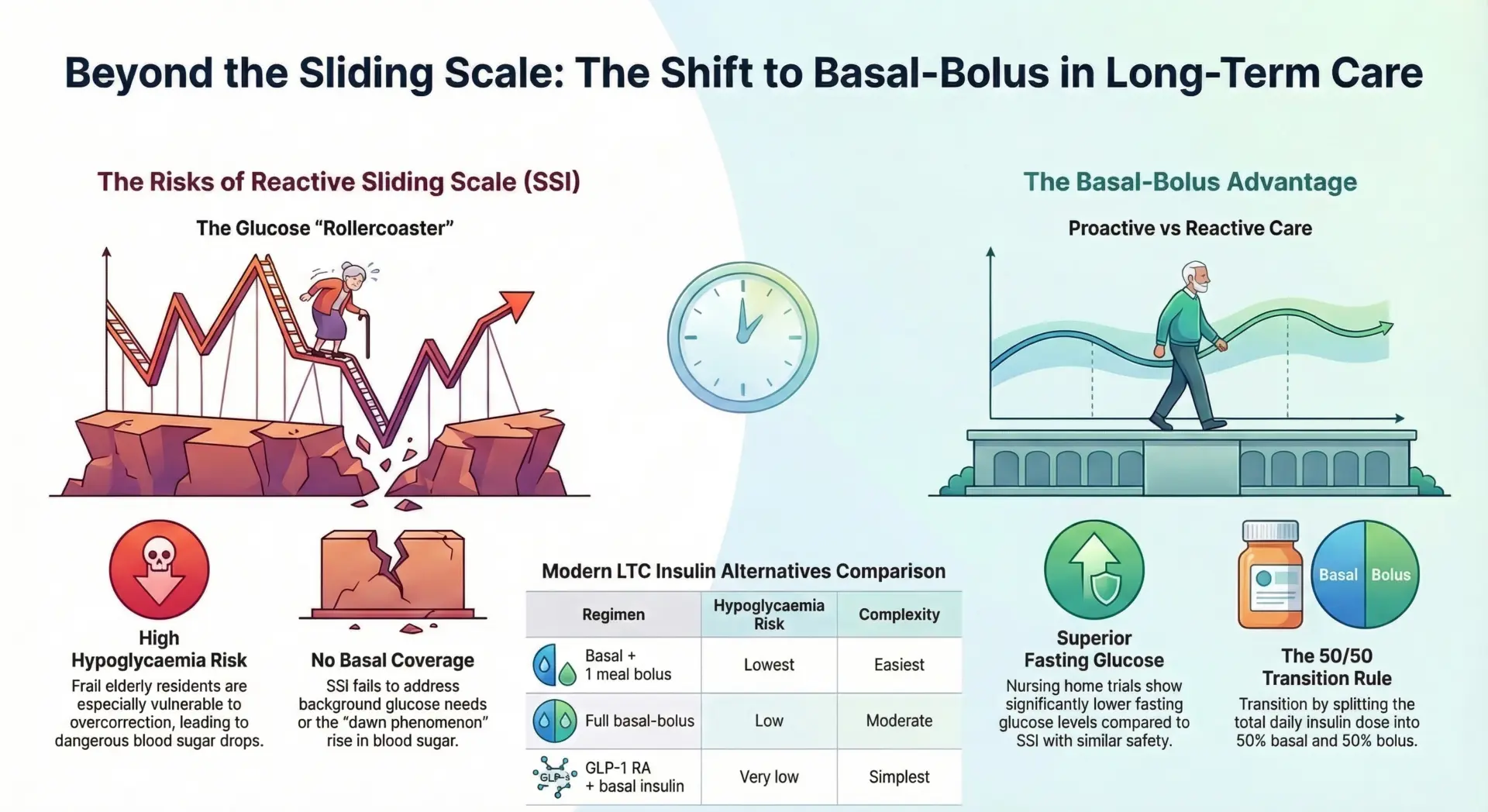
Diabetes = Beta Cell Failure (Different Causes)
Diabetes occurs when the pancreas does not produce enough insulin or the body cannot use it effectively. At its core, every form of diabetes involves beta cell dysfunction but the cause differs dramatically between types.
Type 1 Diabetes (Insulin Dependent Diabetes): Type 1 diabetes is caused by an autoimmune reaction in which the body’s immune system mistakenly attacks and destroys beta cells. By the time of diagnosis, roughly 90% of beta cells have been destroyed. The exact cause of this autoimmune response is not fully understood, though family history and genetic risk factors play a role. Because the pancreas makes little to no insulin, patients require insulin injections including rapid-acting and long-acting formulations or an insulin pump for survival.
Type 2 Diabetes: Type 2 diabetes occurs when the body builds up insulin resistance the cells can no longer respond to insulin properly, prompting the pancreas to pump out more insulin. Over time, beta cells become exhausted. At diagnosis, about 50% of beta cell function is already lost, and this can decline to as little as 10-20% over a decade. Risk factors include obesity, lack of physical activity, poor dietary choices, and family history. Type 2 diabetes can cause high blood glucose levels and prevent the body’s cells from getting enough energy.
| Stage | T1D Beta Cells | T2D Beta Cells | Insulin Therapy |
| Diagnosis | ~10% remaining | ~50% remaining | Injected / oral meds |
| 5 Years | ~5% | ~30% | Pump / intensified |
| 10 Years | <1% | ~10-20% | Pump / transplant? |
Other forms of diabetes include gestational diabetes, which develops during pregnancy and can result in complications during delivery, and diabetes caused by chronic pancreatitis, which can damage the insulin-producing cells in the pancreas. Pancreatic cancer can also impair insulin production.
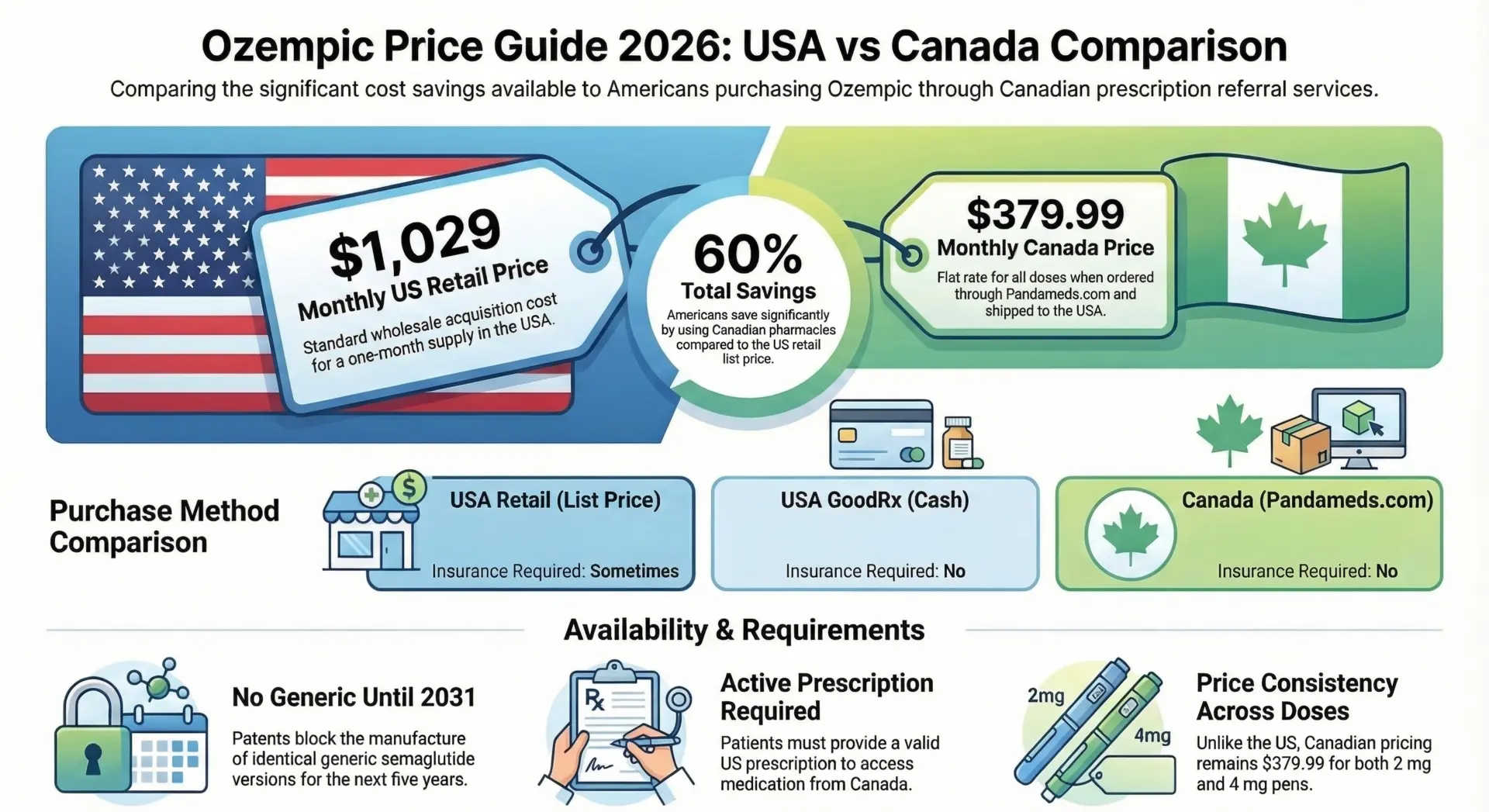
Beta Cells + Ozempic: The GLP-1 Connection
GLP-1 (glucagon-like peptide-1) is a hormone released by the small intestine after eating. It stimulates insulin secretion, suppresses glucagon release, slows gastric emptying, and reduces appetite. The natural hormone is broken down within minutes, but GLP-1 receptor agonists like semaglutide (brand name Ozempic) are engineered to last approximately 7 days, enabling once-weekly dosing.
How Ozempic Works on Beta Cells
Semaglutide binds to GLP-1 receptors on beta cells, increasing intracellular cAMP. This amplifies glucose-dependent insulin secretion meaning the drug only boosts insulin output when blood sugar is elevated, dramatically reducing the risk of low blood sugar (hypoglycemia). It also suppresses glucagon from alpha cells and slows food movement through the stomach.
Clinical Impact:
- A1C reduction of 1.0-1.8 percentage points on average (AAFP)
- Average weight loss of 5-7% of body weight in diabetes trials; the higher-dose formulation (Wegovy) has shown up to 15% weight loss (Harvard Health)
- Reduced risk of major cardiovascular events, including heart attack and stroke
- Evidence of slowed progression of beta cell decline in type 2 diabetes patients
| Without GLP-1 Therapy | With Ozempic (T2D) | Beta Cell Outcome |
| ~50% function lost at diagnosis | Stimulates remaining cells | Slowed decline |
| Progressive exhaustion | Glucose-dependent boost | Extended production window |
T1D Reality: Because type 1 diabetes involves near-complete destruction of beta cells, Ozempic is not effective as a primary treatment there are simply no remaining beta cells to stimulate. The FDA has not approved GLP-1 agonists for type 1 diabetes, though off-label use is being studied.
Anatomy: Where Beta Cells Live in the Pancreas
The islets of Langerhans are scattered throughout the pancreas like tiny islands in a sea of exocrine tissue. Each islet is roughly 50-500 micrometers in diameter about the size of a pinhead and contains approximately 1,000 cells. Key anatomical facts:
- The human pancreas contains roughly 1 million islets, constituting 1-2% of total pancreas volume.
- Beta cells occupy the central core of each islet, making up 50-70% of the islet cell population.
- Islets are highly vascularized, receiving 5-6 mL of blood per minute per gram up to 15 times more blood flow than the surrounding exocrine tissue.
- Insulin released from beta cells enters the hepatic portal vein and travels first to the liver, where it regulates stored glucose release, before circulating to muscles, fat, and other tissues.
The Insulin Pathway: Pancreas → 1 million islets → Beta cells (50-70%) → Insulin granules → Portal vein → Liver → Systemic circulation → Body’s cells.
When beta cells fail, they typically die from the inside out endoplasmic reticulum stress from overwork leads to cell death (apoptosis). This is particularly relevant in type 2 diabetes, where years of insulin resistance force beta cells to work overtime until they burn out.
Insulin Production Problems: T1D vs T2D
Type 1
The immune system attacks and destroys beta cell DNA and cellular machinery, eventually reaching zero insulin production. This autoimmune reaction typically strikes in childhood or young adulthood, though it can occur at any age. Without external insulin (via insulin injections, an insulin pump, or inhaled insulin), blood glucose levels climb dangerously, leading to serious complications affecting the eyes, kidneys, nerves, and heart.
Type 2
A combination of lipotoxicity (fat accumulation in cells) and glucotoxicity (damage from chronic high blood sugar) leads to progressive beta cell exhaustion. Lifestyle factors such as obesity, lack of exercise, and poor dietary choices are major contributors to insulin resistance, which sets the stage for prediabetes and eventually type 2 diabetes. However, insulin resistance can occur in individuals who are not overweight or obese genetics and other risk factors also play a role.
Managing Diabetes: Lifestyle, Diet, and Monitoring
Whether managing type 1 or type 2 diabetes, lifestyle plays a central role in diabetes care. Both types are manageable health conditions when patients work closely with a healthcare provider and make consistent, evidence-based adjustments.
Key strategies include:
- Balanced Diet: Eating a balanced diet with non-starchy vegetables, fruits, whole grains, and lean proteins helps reduce blood sugar levels and supports overall metabolic health.
- Regular Exercise: Physical activity increases insulin sensitivity, helps control blood sugar levels, aids in weight management, and can help reverse insulin resistance.
- Weight Management: Weight loss can improve insulin sensitivity and relieve strain on the body, making it particularly beneficial for managing type 2 diabetes. Even a 5-10% reduction in body weight can significantly improve metabolic health.
- Blood Sugar Monitoring: People with diabetes should regularly check their blood sugar levels to manage their condition effectively. Continuous glucose monitors offer real-time data, while traditional fingerstick tests remain widely used.
- Stress and Sleep: Stress management and getting enough sleep are important lifestyle changes that can help in diabetes management, as both cortisol and sleep deprivation affect blood glucose levels and insulin sensitivity.
- Insulin Therapy: High blood sugars are a common symptom of diabetes, but patients treating their diabetes with insulin injections may occasionally inject too much insulin, causing low blood sugar. Awareness of injection site rotation, proper dosing, and recognizing early signs of hypoglycemia are essential. Allergic reactions at the injection site are rare but should be reported to a healthcare provider.
Beta Cell Replacement Frontiers 2026
The ultimate goal of diabetes research is to restore the body’s ability to produce its own insulin. Several approaches are making significant progress as of 2026:
Stem Cell-Derived Beta Cells: Vertex Pharmaceuticals’ zimislecel (formerly VX-880) is the most advanced program. This therapy infuses stem cell-derived, fully differentiated islet cells into the liver’s portal vein to restore insulin production. In Phase 1/2 data, 10 of 12 patients who received a full dose achieved insulin independence at one year, with over 90% time-in-range for blood glucose. The Phase 3 trial completed enrollment of 50 patients in 2025, with global regulatory submissions expected in 2026.
Islet Transplantation: Traditional cadaveric islet transplants can provide insulin independence for approximately one year, but are limited by donor availability. Roughly 1 million islets are needed per transplant, and there are only about 1,000 pancreas transplants available in the U.S. each year.
Gene Therapy and Regeneration: Researchers are exploring methods to regenerate beta cells in the body or convert other cell types (such as alpha cells) into insulin-producing cells. These approaches remain in early-stage research but represent promising long-term solutions.
GLP-1 agonists like Ozempic function as a bridge therapy buying time by stimulating residual beta cells and improving metabolic health while these more definitive beta cell replacement technologies mature.
Disclaimer: This information is intended for general knowledge and informational purposes only and does not constitute medical advice. Always consult with a healthcare professional for personalized guidance.
Written by the Pandameds.com Editorial Team
Our content is created by pharmacy-trained researchers and healthcare specialists and rigorously reviewed by a diverse panel of authentic experts from the pharmaceutical and healthcare fields. This collaborative review process ensures that every article meets the highest standards of medical accuracy, reliability, and relevance.
- ✅ Authored by pharmacy-trained professionals
- 🔍 Reviewed by multiple verified experts in the pharmaceutical and healthcare niche
- 💊 Based on trusted sources including FDA, Health Canada, and peer-reviewed clinical studies
- 🔄 Regularly reviewed and updated every 90 days to maintain accuracy and trustworthiness
About Pandameds.com
Pandameds.com offers a range of weight loss medications at an affordable price.
Fast, Reliable Shipping to the USA!
Affordable Prescription Meds From Canada
Join our mailing list for exclusive promos, curated health content & more.
Related Blog Posts
Call Us Today!
If you have any questions, please email our support team at support@pandameds.com or call us toll-free at 1-888-862-1210.