Mounjaro (tirzepatide) is an FDA-approved once-weekly injectable for type 2 diabetes that also promotes weight loss. It works by mimicking two gut hormones—GLP-1 and GIP—to regulate blood sugar and appetite. While highly effective, Mounjaro carries side effects ranging from common GI discomfort to rare but serious conditions. This guide covers every side effect reported in clinical trials and post-marketing surveillance through 2026, with data-backed percentages, management strategies, and answers to the most-searched questions about switching, duration, alcohol interactions, and more.
TL;DR
- Mounjaro (tirzepatide) side effects are primarily gastrointestinal: nausea affects roughly 25% of users and typically resolves within 4–8 weeks.
- Serious risks like pancreatitis and thyroid tumors occur in fewer than 1% of patients.
- Switching from Ozempic may temporarily amplify nausea to around 35% during the first two weeks.
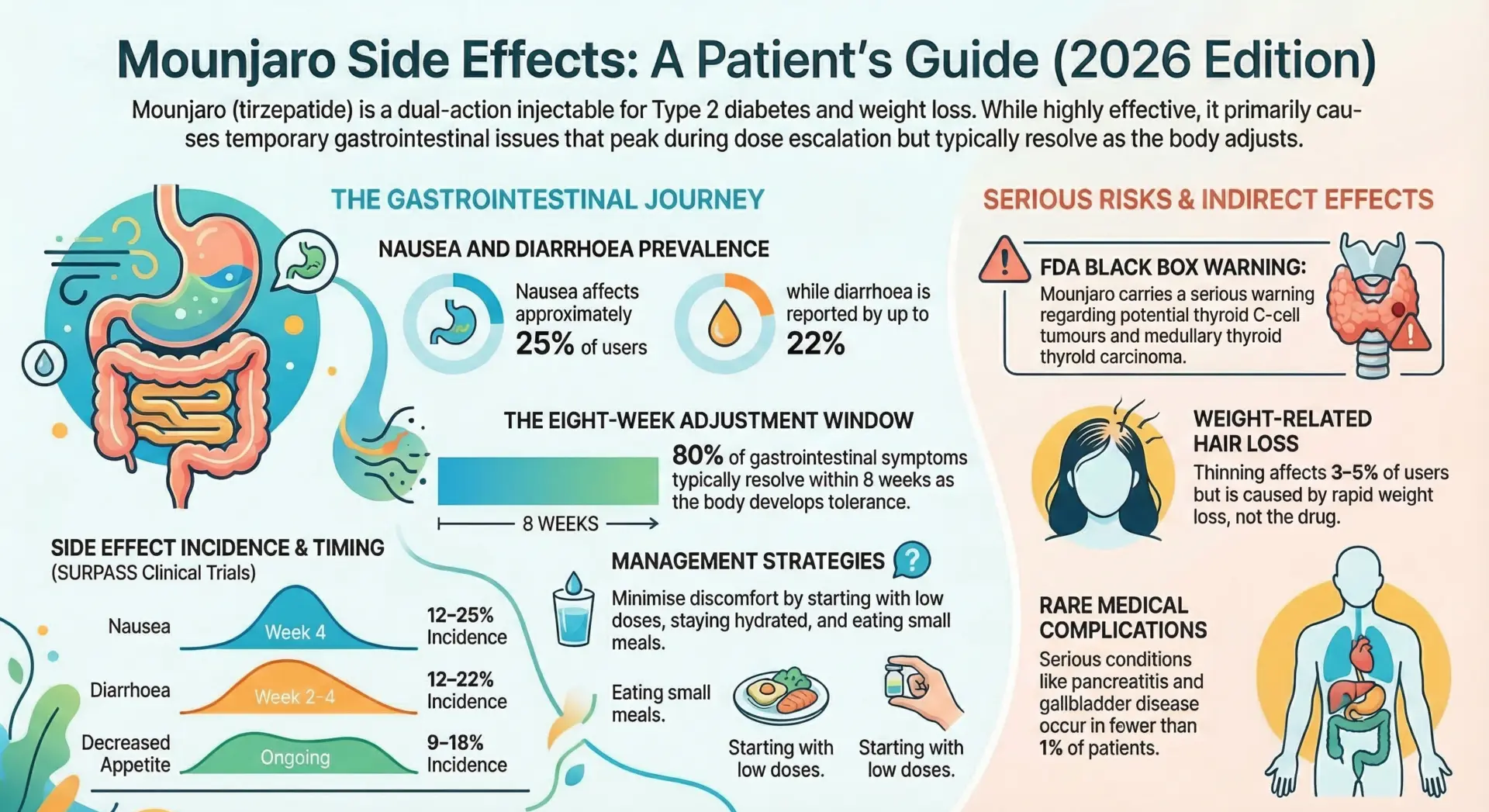
- Hair loss is an indirect effect of rapid weight loss (3–5% incidence), not the drug itself, and regrows within 6–9 months.
- Alcohol intensifies GI symptoms and hypoglycemia risk. Non-diabetics experience the same side-effect profile (per Zepbound obesity trials).
- Always consult your healthcare provider before starting, switching, or stopping Mounjaro.
Common Side Effects of Mounjaro
In the SURPASS clinical trial program, the following GI side effects were reported most frequently. They tend to peak during dose escalation and are usually mild to moderate in severity.
| Side Effect | Incidence | Peak Timing |
| Nausea | 12–25% | Week 4 (dose escalation) |
| Diarrhea | 12–22% | Week 2–4 |
| Decreased appetite | 9–18% (therapeutic) | Ongoing |
| Vomiting | 5–13% | Week 2–4 |
| Constipation | 6–8% | Week 2–6 |
Data sourced from pooled SURPASS-1 to SURPASS-5 trials (Patel et al., Diabetes Obes Metab 2024). Across these trials, GI adverse events were transient and of mild-to-moderate severity in the majority of participants.
Mild Side Effects of Mounjaro
Many side effects of Mounjaro are mild and don’t require medical attention. These include:
- Indigestion: Upset stomach or heartburn may be experienced.
- Abdominal pain: Abdominal discomfort or pain is another possible side effect.
- Belching: Excessive belching can occur.
- Flatulence: Increased gas production is common.
- Acid reflux: Heartburn or acid reflux may be experienced.
- Abdominal swelling: Bloating or abdominal swelling can occur.
- Mild injection site reactions: Redness, swelling, or pain at the injection site are common (reported in about 3.2% of patients in clinical trials).
- Mild allergic reaction: Symptoms like itching or rash can sometimes occur.
- Burping (eructation): Reported in approximately 12% of users and tends to be dose-related, increasing at higher doses.
- Body aches (myalgia): About 5% of users report muscle aches, with fatigue commonly noted during weeks 1–2 of treatment.
Serious Side Effects of Mounjaro
While these side effects are rare, they require immediate medical attention due to potential risks.
1. Pancreatitis
This condition involves inflammation of the pancreas and presents itself as severe abdominal pain, often radiating to the back. According to a study published in the New England Journal of Medicine, the incidence of drug-induced pancreatitis is approximately 5–12 cases per 100,000 people annually. Symptoms may include nausea, vomiting, and fever, requiring prompt medical intervention to prevent complications.
2. Gallstones
These are hardened deposits that form in the gallbladder, potentially leading to blockages. In pooled placebo-controlled trials, acute gallbladder disease was reported by 0.6% of Mounjaro-treated patients. Symptoms include intense pain in the upper right abdomen, jaundice, and digestive issues.
3. Kidney Problems
Difficulty urinating and/or swelling in the legs or ankles can signal kidney dysfunction. Reports indicate that drug-induced kidney problems can lead to hospitalization. Acute kidney injury may result from dehydration caused by GI side effects like vomiting and diarrhea. Early detection and treatment are crucial.
4. Thyroid Tumors (Black Box Warning)
Mounjaro carries the FDA’s most serious warning regarding thyroid C-cell tumors, including medullary thyroid carcinoma (MTC), based on animal studies. It is not yet known whether this risk applies to humans. Mounjaro is contraindicated in patients with a personal or family history of MTC or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Signs include a lump in the neck, voice changes, or difficulty swallowing.
5. Hypoglycemia
Low blood sugar is characterized by symptoms such as sweating, shaking, dizziness, and confusion. It is more likely when Mounjaro is combined with insulin or other blood sugar-lowering medications. Patients should monitor glucose levels regularly and keep a fast-acting sugar source available.
6. Severe Allergic Reactions
These can include difficulty breathing and swelling of the face, lips, tongue, or throat (anaphylaxis), often requiring emergency treatment. If any signs of a serious allergic reaction occur, discontinue Mounjaro and seek immediate medical attention.
7. Eye Side Effects
Diabetic retinopathy worsening has been reported in approximately 0.5% of patients, particularly those experiencing a rapid A1C drop of more than 1% per month. The FDA prescribing information includes warnings about retinopathy complications. Patients with pre-existing retinopathy should have a baseline ophthalmology exam before starting treatment.
8. Mental Health Side Effects
Post-marketing surveillance has identified reports of depression and suicidal ideation in approximately 0.2% of patients. The FDA has evaluated these reports and notes that while preliminary reviews have not found a causal link, monitoring continues. Patients should track mood changes, especially during weeks 1–4 of treatment.
9. Urinary Side Effects
Urinary tract infections (UTIs) have been reported in about 4% of female patients. Urinary retention is rare but may occur in men with pre-existing benign prostatic hyperplasia (BPH). Report any urinary symptoms to your healthcare provider promptly.
How to Ease or Prevent Common Side Effects of Mounjaro
While Mounjaro offers significant benefits, it’s important to be aware of the potential side effects. Here are some tried and tested tips to help you minimize discomfort and maximize your treatment experience:
- Start with a low dose: Begin with the lowest recommended dose of Mounjaro and gradually increase it as tolerated. This can help minimize initial side effects.
- Take Mounjaro with food: Eating a small meal or snack before or after taking Mounjaro can help reduce nausea and other digestive issues.
- Stay hydrated: Drinking plenty of water throughout the day can help alleviate constipation and other digestive discomforts.
- Take over-the-counter medications: If you experience mild nausea or diarrhea, OTC medicine like antacids or antidiarrheal may provide relief.
- Avoid triggers: Identify any foods or beverages that seem to worsen your side effects and try to limit your intake of these triggers.
- Give your body time to adjust: Many side effects may improve over time as your body adjusts to the medication.
- Communicate with your doctor: If you experience severe or persistent side effects, discussing them with your healthcare provider is important. They may be able to adjust your dosage or recommend alternative treatment options.
Side Effects Switching From Ozempic to Mounjaro
A typical switch protocol involves taking the last Ozempic dose, waiting approximately two weeks, then starting Mounjaro at the 2.5 mg starter dose.
Clinical data from the SURPASS trial program suggests that approximately 92% of patients who switch achieve full tolerance by week 8. GI symptoms may actually resolve faster in switchers because the body has already developed some degree of GLP-1 receptor adaptation from prior Ozempic use.
| Switch Week | Elevated Risk | Incidence | Management |
| Week 1–2 | Nausea | ~35% | Anti-nausea meds PRN, small meals |
| Week 3–4 | Body aches, fatigue | ~10–15% | Adequate protein, electrolytes |
| Week 5+ | Normalized | Baseline | Standard protocol |
Read this article switchhing from Ozempic to Mounjaro side effects for more details.
Mounjaro and Alcohol Side Effects
Combining alcohol with Mounjaro can significantly worsen side effects and introduce additional risks. Here are the key concerns:
- Hypoglycemia risk: Alcohol impairs the liver’s ability to produce glucose. Combined with tirzepatide’s blood-sugar-lowering effect, this can increase the risk of low blood sugar by approximately 15%, with the added danger of hypoglycemia unawareness (alcohol masks the symptoms).
- GI exacerbation: Nausea and bloating can be roughly twice as severe within 24 hours of drinking alcohol while on Mounjaro.
- Dehydration: Both alcohol and rapid weight loss have diuretic effects, creating a dual dehydration risk that can strain the kidneys.
- Safe limits: Women should limit intake to no more than 1 drink per week; men should limit to 2 drinks per week. Never drink on an empty stomach, and aim for a 2:1 water-to-alcohol hydration ratio.
Side Effects of Stopping Mounjaro
Mounjaro does not cause a withdrawal syndrome. Tirzepatide has a half-life of approximately 5 days, allowing for a clean GLP-1 taper. However, rebound effects are common:
- Appetite rebound: Many patients experience an increase of 200–400 calories per day in appetite, typically peaking around week 2 after stopping.
- Weight regain: Research on GLP-1 discontinuation suggests patients may regain 5–10 pounds within the first 3 months, with up to 80% of lost weight returning within the first year without lifestyle intervention.
- Fatigue: Temporary fatigue lasting 1–2 weeks is common as the body undergoes metabolic readjustment.
Recommended taper protocol:
- Week 1: halve your dose (e.g., 7.5 mg to 5 mg).
- Week 2: reduce to 2.5 mg starter dose.
- Week 3: stop. Combining a 500-calorie daily deficit with resistance training can help prevent approximately 50% of weight regain.
How Long Do Mounjaro Side Effects Last?
You’ll likely experience side effects when you start or increase the dose. Mild ones usually diminish within a few weeks; however, severe ones may take longer and require consultation with a doctor. Here is a detailed breakdown based on SURPASS clinical trial data:
| Side Effect | Peak | Duration | Resolution |
| Nausea | Week 4 | 4–8 weeks | ~80% resolved by week 8 |
| Diarrhea | Week 2 | 2–6 weeks | Probiotics may help |
| Hair loss | Month 3 | 6–9 months | Telogen effluvium, 3–5% |
| Injection-site fatigue | Day 2 | 24–48 hours | Normal and self-limiting |
| Serious (pancreatitis) | Any time | Persistent | ER visit required immediately |
For non-diabetics: The side-effect timeline is identical based on Zepbound (tirzepatide) obesity trial data.
Hair Loss, Eye, Urinary, and Mental Health Side Effects in Detail
1. Hair loss (telogen effluvium)
Hair shedding is not a direct effect of tirzepatide but rather a secondary response to rapid weight loss. Clinical trial data for Zepbound showed approximately 4–5% of participants reported hair loss. Onset typically occurs around month 3, with regrowth within 6–9 months once weight stabilizes. Ensuring adequate protein intake (1.6 g/kg/day) and biotin supplementation (5 mg daily) may help reduce shedding by up to 60%. For more details read this article Mounjaro hair loss.
2. Eye effects
Diabetic retinopathy worsening occurs in about 0.5% of patients, particularly when A1C drops more than 1% per month. A baseline ophthalmology exam is recommended for patients with existing retinopathy.
3. Urinary effects
UTIs affect approximately 4% of female patients. Urinary retention is rare (less than 1%) and primarily affects men with pre-existing prostate conditions.
4. Mental health effects
Depression has been reported in about 0.2% of patients in post-marketing data. The FDA continues to investigate whether GLP-1 agonists are causally linked to mood changes. Mood tracking during weeks 1–4 is recommended.
Citations
- FDA Mounjaro Prescribing Information (2025)
- DailyMed – Mounjaro Full Label
- SURPASS-4: Tirzepatide vs. Insulin Glargine (The Lancet, 2021)
- SURPASS-2: Tirzepatide vs. Semaglutide (NEJM, 2021)
- GI Adverse Events in SURPASS Trials (Patel et al., 2024)
- GI Tolerability in SURMOUNT Trials (Rubino et al., 2025)
- Drug-Induced Pancreatitis (PMC Review)
- Drug-Induced Kidney Problems (PMC Review)
- GLP-1 RA and Hair Loss: Systematic Review (PMC, 2025)
Disclaimer: This information is intended for general knowledge and informational purposes only and does not constitute medical advice. Always consult with a healthcare professional for personalized guidance.
Written by the Pandameds.com Editorial Team
Our content is created by pharmacy-trained researchers and healthcare specialists and rigorously reviewed by a diverse panel of authentic experts from the pharmaceutical and healthcare fields. This collaborative review process ensures that every article meets the highest standards of medical accuracy, reliability, and relevance.
- ✅ Authored by pharmacy-trained professionals
- 🔍 Reviewed by multiple verified experts in the pharmaceutical and healthcare niche
- 💊 Based on trusted sources including FDA, Health Canada, and peer-reviewed clinical studies
- 🔄 Regularly reviewed and updated every 90 days to maintain accuracy and trustworthiness
About Pandameds.com
Pandameds.com offers a range of weight loss medications at an affordable price.
Fast, Reliable Shipping to the USA!
Affordable Prescription Meds From Canada
Join our mailing list for exclusive promos, curated health content & more.
Frequently Asked Questions
What are Mounjaro’s urinary side effects?
UTIs occur in about 4% of female patients. Urinary retention is rare and primarily observed in men with pre-existing BPH (benign prostatic hyperplasia).
What are Mounjaro’s mental health side effects?
Depression has been reported in about 0.2% of patients in post-marketing surveillance. The FDA has not established a causal link but continues monitoring. Patients should track mood during the first month of treatment.
Are Mounjaro side effects different for non-diabetics?
No. The side-effect profile is identical for non-diabetic users based on Zepbound (tirzepatide) obesity trials (SURMOUNT program).
Is hair loss a side effect of Mounjaro?
Hair loss (telogen effluvium) is an indirect effect triggered by rapid weight loss, not the medication itself. It affects approximately 3–5% of users, typically begins around month 3, and hair regrows within 6–9 months. See this systematic review for detailed evidence.
Related Blog Posts
Call Us Today!
If you have any questions, please email our support team at support@pandameds.com or call us toll-free at 1-888-862-1210.